The pace of change within the biotechnology sector is growing at a great rate and the perfusion bioreactor has been one of the greatest innovations contributing to the change. There is an advantage to this system because it provides an effective alternative to the conventional batch and fed-batch culture, giving cells the ability to be cultured continuously in a high productiveness and viability manner over long intervals.
So why is this bioreactor special in current bioprocessing? What is behind the move to this system by pharmaceutical giants and research laboratories in production of vaccines, the manufacturing of antibodies, and in cell therapy?
Well, it is time to go into the ways perfusion bioreactors are transforming the possibilities of the future of biotechnology.
Understanding the Concept
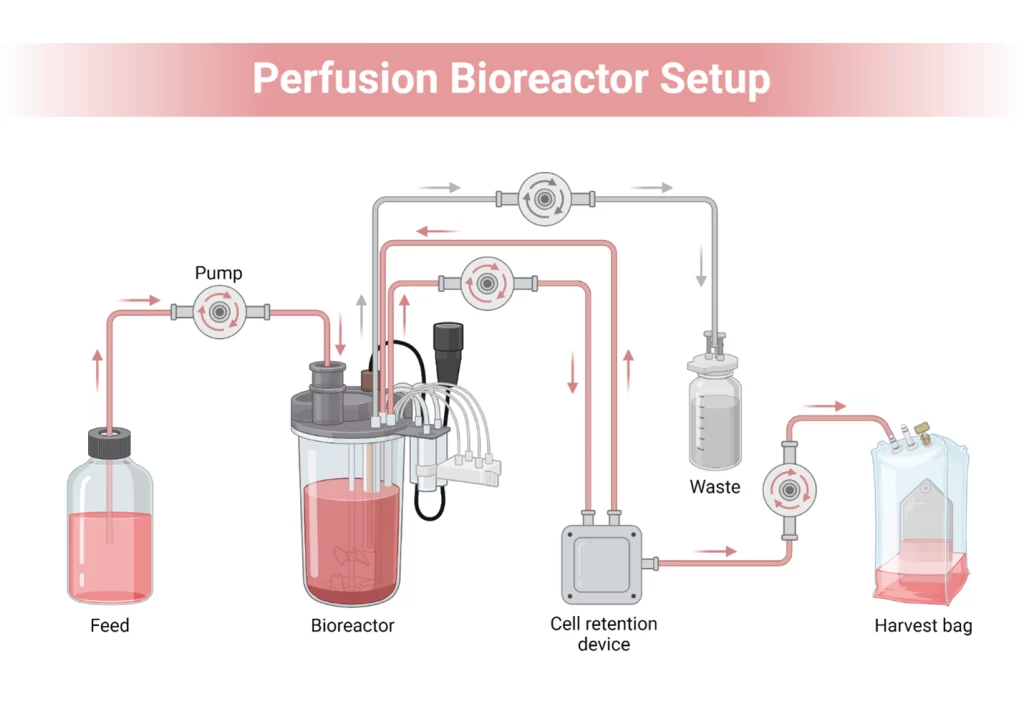
Perfusion bioreactor Perfusion bioreactor is a type of cell culture system in which fresh media is perfused in and waste products are percolated out, but at the same time the cells remain confined within the bioreactor. This leaves a rigorous high concentration of culture that is preferable in long term production runs.
In contrast to the technique of harvesting of cells that follows upon one cycle in a batch system, the perfusion system in bioreactors permits continuous process that may continue up to the satisfaction of several weeks or even months.
Key characteristics of perfusion culture:
- Continuous nutrient supply
- Consistent removal of toxic by-products
- High cell density and product yield
- Steady-state operating conditions
- Minimal shear stress for sensitive cells
It makes them particularly appropriate for production of biologics, production of monoclonal antibodies, stem cell growth and development of viral vaccines.
Still wondering how it works? Let’s explore the components and mechanics next.

Components and Working
An intelligent design makes implemented this bioreactor successful. It incorporates a standard bioreactor chamber; cell retention solutions and media control droops which enable continuous in-and-out exchange without the loss of precious cells.
Core components:
- Bioreactor vessel (stirred-tank, wave, or hollow fiber-based)
- Cell retention device (TFF membrane, spin filter, acoustic separator)
- Media reservoir (fresh feed and waste collection)
- Pump systems (peristaltic or diaphragm pumps)
- Sensors and control units (pH, DO, glucose, lactate)
How it works:
- The reactor is filled constantly with fresh media.
- The particles are retained by retention device and spent media and metabolites to go through.
- Waste is drained out constantly with the environment kept nutrient-rich with an optimal growth environment to the cells.
Such an effective arrangement allows cultures to grow and generate biologics without encountering frequent disturbances as they do during batch procedures.
Типы
Different biotechnology applications call for different bioreactor types. This bioreactor is very flexible and can take a numerous number of forms to suit particular purposes.
Common types include:
Stirred-Tank Perfusion Bioreactors
Synonymous with tests in labs and industry and spin filters or TFF modules are added to retrieve.
Hollow Fiber Bioreactors (HFB)
Applied to tissue engineering where cells are allowed to grow as bundles of fiber and nutrients diffuse in the pores.
Wave Perfusion Bioreactors
Rocking systems that can be used in perfusion of sensitive mammalian cells are disposable.
Acoustic Perfusion Bioreactors
Special cells can be retained using sound waves thus reducing clogging and shear.
Fixed Bed or Packed Bed Bioreactors
It is typical in cell therapy where the cells cultivate on micro carriers within packed columns.
Both of them have their advantages regarding the cell line, scale, product, and cost. The case in point is that the HFBs are ideal in case of stem cells, whereas the acoustic systems are excellent in mAb production using CHO cells.
Преимущества
What makes perfusion bioreactor so popular to biotechnology specialties? The benefits go beyond just productivity.
Major advantages:
Increased yield per unit volume
The load of the cells would be greater and the volume in the reactor could be less.
Better product quality
Minimized cell stress leads to the regular glycosylation and protein folding.
Непрерывное производство
No downtime between batches; ideal for commercial-scale biologics.
Efficient nutrient utilization
Continuous feed avoids depletion or excess accumulation.
Ideal for sensitive or slow-growing cells
Such as hybridisms, stem cells, or primary cells.
Масштабируемость
Perfusion is able to scale to bench-top apparatus through to commercial 2000L reactors in a modular fashion.
Consider how 10-fold more product can be made with the same size bioreactor. That’s the promise of these bioreactors , efficiency at its finest.
Applications in Biopharmaceuticals and Beyond

The potential application of the this bioreactor will be in many areas, but there will be possibilities on biopharma, in particular.
Key applications:
Monoclonal Antibody Production
High titers are obtained with CHO cells in perfusion culture as compared to those in fed-batch modes.
Производство вакцин
Influenza, COVID-19 or adenovirus virus production is more consistent in viral vectors.-
Cell and Gene Therapy
Expansion of stem cells, T-cells or iPSCs under GMP with high level of purity.
Recombinant Protein Production
Human proteins, enzymes, and hormones with better quality control.
Tissue Engineering
Supporting 3D cell constructs in perfusion for regenerative medicine.
Research and Development
Used in long-term experiments and pharmacokinetic studies.
This renders these bioreactor as a revolutionary hand in both academia and industrial bio-processing laboratories.
Optimization Strategies for Performance
Operating a perfusion bioreactor is both art and science. A number of strategies have to be refined depending on a process and product to maximize output.
Tips for optimal operation:
Control perfusion rate
When it is too fast, cells can be washed out, when being too slow enough, waste builds up.
Monitor nutrient and waste levels
The level of glucose, glutamine, lactate, ammonia, and pH should also be within optimal levels.
Use high-efficiency retention devices
Ensure minimal clogging and maximum cell recovery.
Maintain steady temperature and DO
Fluctuations reduce productivity and stress cells.
Adapt feeding strategies
Add supplements like lipids or hydrolysates for high-yield cultures.
Automate data collection
Observe the changes and anticipate them with the help of real-time analytics and AI.
Process analytical technology (PAT) tools are increasingly integrated into perfusion platforms to ensure quality by design (QbD) compliance.
Проблемы и ограничения
While promising, perfusion bioreactor systems aren’t without hurdles.
Common challenges:
Расходы
Initial configuration and filters/pumps also put it above the price scale of batch systems.
Complexity
Continuous monitoring and control require skilled personnel.
Риск загрязнения
The higher run times predispose the higher exposure and hence aseptic handling is imperative.
Filter Fouling
Membranes can clog over time, especially with high-density cultures.
Scale-up Issues
At higher volumes the flow rates, oxygenation and waste removal are difficult to balance.
Nevertheless, technological advances such as disposable perfusion bags, modular systems, and internet-based sterilization guidelines are making up for them.
Future Trends in This Bioreactor Technology
The biotechnology sector is pouring huge investment in high-end perfusion bioreactor systems. Our future systems are expected to be more intelligent, swift, automated as we venture into the world of personalized medicine and highly demanded biologics.
What’s next?
- AI-integrated bioreactors for predictive monitoring and control
- Disposable perfusion systems with built-in sensors
- 3D printed perfusion chambers for organoid and tissue models
- Modular bio manufacturing pods for decentralized production
- Hybrid systems combining perfusion and fed-batch features
Continuous bioprocessing (perfusion) will grow to be the conventional approach at the many therapeutic pipelines as well as upstream bottlenecks move downstream.

Заключение
Perfusion bioreactor technology represents far more than just a marginal improvement or a simple “technological step-up.” It is a paradigm changer and transforms altogether the biopharmaceutical production.
Given the relentless increase in global demand for both complex biologics (such as monoclonal antibodies, vaccines, and recombinant proteins) and groundbreaking cell therapies (like CAR-T cells and other regenerative medicines), the limitations of traditional manufacturing methods are becoming increasingly apparent. The ability to produce at high density, continuously, and at a high rate of output is an advantage of perfusion systems whose forthcoming need can easily be seen.
They will be ready to attend to the growing demand of patient populations all across the globe, accelerate the designing and manufacture of next-generation therapeutics, and optimize the supply chain, providing the biotechnology sector with a promise to safely supply life-saving and life-altering therapies well into its future.
Looking to elevate your bioprocessing setup? Explore cutting-edge perfusion bioreactor solutions tailored for your industry needs at БайЛун Био and transform your productivity today!
Часто задаваемые вопросы
What is this bioreactor used for?
It has been applied in continuous cell culture and this enables steady production of biologics such as antibodies, proteins or viral vectors.
Which cells are grown in perfusion bioreactors?
The cell lines are frequently used: CHO cells, HEK293, hybridomas, stem cells and primary mammalian cells.
Is perfusion better than batch culture?
Of course, to high-value biologics, perfusion yields higher quantities with higher quality and continuous production.
Do perfusion systems work for stem cell culture?
Absolutely. They have a stable, low shear, nutrient perfect environment to permit stem cell expansion.
Can perfusion bioreactors be scaled up?
Yes. With good design they are able to scale between 1L on the benchtop up to 2000L GMP reactor.
How are cells retained in perfusion systems?
Using membranes, spin filters, acoustic separators, or gravity-based methods.
Are perfusion systems GMP compliant?
In regulated environment, modern systems (that have reasonable control modules) are fully compliant and deployed with high frequency.


