This paper studies the synthetic biomanufacturing of propionic acid. It reviews research progress in biomanufacturing in detail. It discusses metabolic engineering of propionibacteria. It also discusses pathway construction in non-native hosts. It further describes bioconversion production using Pseudomonas. The paper gives technical routes and strategies for sustainable propionic acid production.
Importance of Propionic Acid and Limits of Traditional Synthetic Methods
PA is an important C3 platform compound, and it is used in many fields. Traditional chemical production uses petroleum as the raw material, and this process causes environmental pollution. This method is also not sustainable.
Microbial fermentation is often used as another method. However, when propionibacteria are used, several problems occur. PA inhibits bacterial growth, so cell growth is slow. The fermentation time is long, and many by-products are produced. In addition, there are few efficient tools for genetic modification.
Research Progress in Biomanufacturing of Propionic Acid
- Metabolically Engineered Propionibacteria for Fermentative Production
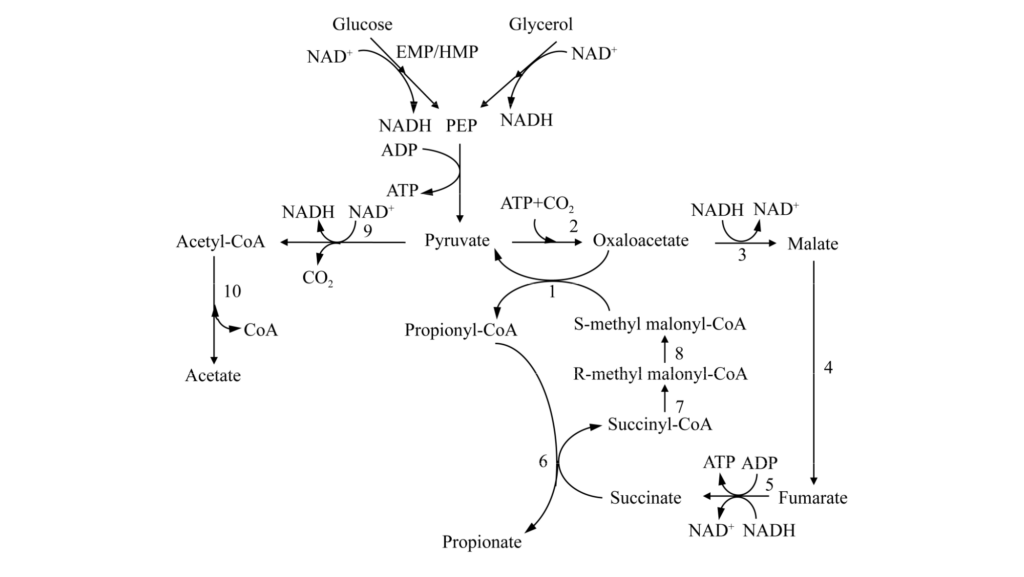
Propionibacteria can use many low-cost carbon sources, so they are suitable for fermentation. Scientists use genetic engineering to improve substrate use. For example, researchers overexpress related genes in Propionibacterium freudenreichii, so the strain can use xylose. In other studies, engineered strains are modified to improve carbon use efficiency.
These bacteria can co-ferment glycerol and glucose. These methods can reduce costs. In the future, combined fermentation of glycerol and cellulose hydrolysates may further lower costs.
Enhance synthetic capacity: It is also important to improve the fermentation performance of the bacteria themselves. Scientists use methods like genome recombination. They rebuild the acrylate pathway. They optimize the expression of key enzymes. Or they make the bacteria express foreign enzymes. These methods can increase propionic acid output.
Enhance acid stress resistance: Improving the bacteria’s tolerance to propionic acid is key. Scientists have found acid-resistant factors. These include the arginine deiminase system and glutamate decarboxylase system. They modify related components. Or they use fiber-bed bioreactors. They also use cell immobilization. These methods can improve the bacteria’s acid resistance. They can also increase propionic acid production.

Reduce by-products: Scientists regulate redox balance. They overexpress related enzymes. These ways can reduce the formation of by-products like acetic acid. They can improve the ability to synthesize pa.
- Metabolically Engineered Non-native Hosts for Propionic Acid Production
Build propionic acid-producing Escherichia coli strains: Escherichia coli grows and reproduces quickly. These advantages make it a research target. But wild bacteria do not have natural enzymes for synthesizing it. Scientists modify them through many ways. They introduce the acrylate pathway. They make the bacteria express the Wood-Werkman cycle from other organisms. They activate the sbm operon. Now, the it output of Escherichia coli has increased a lot. But more optimization is still needed.
Build propionic acid-producing Saccharomyces cerevisiae strains: Saccharomyces cerevisiae is acid-tolerant. It is also biologically safe. Scientists enhance L-threonine synthesis. They integrate it into the propionic acid synthesis pathway. These modifications let it produce a certain amount of it under aerobic fermentation. It may become a potential host.

- Pathway Design and Optimization for Bioconversion of Propionic Acid
L-threonine conversion route
Modify native pathways: Pseudomonas putida KT2440 can synthesize high-purity it. It does this by modifying the L-threonine degradation pathway. Scientists knock out genes in branched metabolic pathways. They integrate transporter genes. They replace promoters. They introduce foreign genes. They knock out propionyl-CoA synthetase. These steps help achieve high molar conversion rate of it. They also found the main thioesterase responsible for propionic acid production. They found key factors for propionic acid tolerance.
Design and optimize artificial pathways: To overcome the coenzyme A limitation in native pathways, scientists designed new pathways. They introduced a decarboxylase from Lactococcus lactis. This enabled more efficient propionic acid production. They established a process. It directly uses L-threonine fermentation broth to convert into propionic acid. This reduces costs. They also identified the aldehyde dehydrogenase that oxidizes propanal in Pseudomonas putida KT2440.
1,2-propanediol conversion route
Using 1,2-propanediol to produce it has the advantage of high atom economy. Scientists screen glycerol dehydratase. They finely regulate its expression. They combine this with the native oxidation ability of Pseudomonas putida KT2440. This achieves efficient conversion of 1,2-propanediol to propionic acid. The titer is slightly lower than the L-threonine route. But the yield is high. In the future, optimization can improve the titer. It is also necessary to solve the problem of vitamin B₁₂ supply for glycerol dehydratase.
Levulinic acid conversion route
Pseudomonas putida EM42 can use levulinic acid to produce propionic acid. Scientists delete related genes. They express foreign genes. These modifications increase propionic acid output. But the molar conversion rate and theoretical mass yield need to be improved.

О Bailun
Bailun has rich experience in the manufacturing of various bioreactors and pressure vessels. It has an expert team integrating biological reaction, fermentation technology, mechanical manufacturing and automatic control. Its scientific research and technological level maintains a leading position in China and first-class international standards, providing you with comfortable, reliable and reassuring product experience. Связаться с нами


