Abstract
Bioreactor technology is widely used in cell culture-based viral vaccine production. Especially during the COVID-19 pandemic, its development and application enabled efficient and low-cost vaccine manufacturing to meet global vaccine demand.
Starting from the classification of viral vaccines and animal cell lines used in vaccine production, this paper introduces the main types of bioreactors, summarizes key parameters related to large-scale viral amplification and their effects on virus yield, systematically evaluates process optimization strategies for producing different viruses (SARS-CoV-2, influenza, tropical viruses, enteroviruses, and rabies virus) using bioreactors, and discusses the synergistic development of bioreactors and computational biology. This review aims to provide valuable information for laboratory-scale simulation and industrial large-scale vaccine production.

1. Introduction
Bioreactors play a crucial role in large-scale cell culture-based viral vaccine production (Figure 1). With advances in vaccine manufacturing, increasingly scalable bioreactors and virus-susceptible cell lines have been applied to produce various vaccines.
In 1962, Capstick et al. adapted BHK21 cells to suspension culture and applied them to veterinary vaccine production. In 1967, Van Wezel developed microcarriers, enabling adherent cell culture in bioreactors, marking the beginning of large-scale cell culture.
After the 1980s, the suspension culture of CHO cells was established. The development of therapeutic antibody production greatly promoted the application of bioreactors in the biopharmaceutical industry. By the end of the 20th century, bioreactor scales had reached 10,000 liters. Currently, with the development of fed-batch, perfusion, and genetic engineering technologies, bioreactors have become platforms for producing viral vectors, live viruses, and virus-based vaccines. Combining the low-shear environment of traditional systems and the scalability of automated systems, they show broad application prospects.
Emerging infectious diseases severely threaten social stability and public health. In particular, coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has spread globally.
Facing the pandemic, vaccine development accelerated at an unprecedented rate. As of April 4, 2022, 11.25 billion vaccine doses had been administered worldwide. Public health experts estimated that a very high vaccination rate against SARS-CoV-2 is required to induce herd immunity. However, the pandemic devastated the economies of many low-income countries, limiting funding for vaccine production and procurement and resulting in unequal global vaccine distribution.
In addition, mRNA vaccine production faces bottlenecks such as shortages of raw materials and difficulties in scaling up. Traditional inactivated whole-virus vaccines are easier to distribute and store (requiring only refrigeration), but their yields cannot meet herd immunity demands.
For most pathogens similar to SARS-CoV-2, large-scale vaccine production is critical for global disease control and eradication, as the high mutation rate of viruses may reduce vaccine efficacy. Therefore, research on viral vaccine production and culture, especially at the laboratory scale, is urgently needed.
Cell culture using bioreactors to produce antigens, antibodies, and other products is a core technology in the large-scale manufacturing of biological products. Precise process control combined with the screening and adaptation of high-expression cell lines can improve production efficiency and product quality while reducing costs.
Laboratory-scale experiments allow optimization of routine physical and chemical parameters, enabling the development of robust culture protocols before industrial scale-up and saving subsequent time. More importantly, laboratory-scale research provides pilot conditions for the development of new biological products, especially novel vaccines, to maximize yield and quality.

2. Bioreactor-Based Vaccine Production
2.1 Vaccine Types
Among various vaccines, mainstream viral vaccines can be broadly divided into two categories: inactivated vaccines and live attenuated vaccines. Within this classification, several subtypes can be produced using different technical approaches, including protein subunit vaccines, virus-like particle vaccines, replicating viral vector vaccines, and nucleic acid vaccines (Figure 1C).
Protein subunit vaccines: Based on synthetic peptides or recombinant proteins, using viral proteins or fragments to induce immune responses.
Virus-like particle vaccines: Contain viral proteins that mimic viral structure but lack genetic material.
Viral vector vaccines: Use non-replicating viruses to deliver DNA containing viral genes into human cells.
Inactivated vaccines: Cultivate viruses in continuous cell lines or tissues, followed by purification, concentration, and inactivation.
Live attenuated vaccines: Reduce virulence by deleting specific viral components or using codon optimization while retaining immunogenicity.
When viruses spread and mutate, new strains may emerge that evade existing vaccines, altering the protective efficacy of different vaccine types. Viruses with high mutation rates are unsuitable for live attenuated vaccines, as mutations may revert during production.
In addition, replication of attenuated strains in cell culture may decrease, leading to lower process yields. In contrast, inactivated vaccines prepared with wild-type viruses yield higher quantities, but highly pathogenic wild-type live viruses may require biosafety level 3 conditions. Screening and adaptation of high-expression cell lines, as important components of large-scale cell culture-based vaccine production, are introduced in the next section.
2.2 Selection and Development of Cell Lines
The development of cell culture systems for viral propagation has advanced viral vaccines. Primary chicken embryo fibroblasts are commonly used in human vaccine production. Currently, continuous cell lines overcome the limitations of primary cells and adapt to modern culture techniques, with “designed cell lines” primarily used to enhance viral yields. These engineered cells are well-characterized and serve as specific cell substrates.
The Vero cell line proliferates only with suitable microcarrier surfaces. Preflucel®, a seasonal inactivated influenza vaccine produced by Baxter using Vero cells, was approved in the EU in 2010. It comprises H1N1, H3N2, and influenza B strains, with Vero cells grown on Cytodex 3 microcarriers.
A recent study using vesicular stomatitis virus (VSV) as a model found that suspension-adapted Vero cells in serum-free medium (SFM) achieved maximum cell densities similar to or better than those in commercial SFMs, with higher densities (8×10⁶ cells/mL) in IHMO3 medium.
Madin-Darby canine kidney (MDCK) cells have become the major suspension cell line for influenza vaccine production. Currently, suspension MDCK cells are amplified in bioreactors with alternating tangential flow (ATF) perfusion systems, with peak influenza A virus titers reaching 4.37 log₁₀ hemagglutination units (HAU)/100 μL and infectious titers of 1.83×10¹⁰ viral particles/mL.
PER.C6 cells can rapidly reach high densities (up to 10⁷ cells/mL) in SFM without solid supports and are susceptible to all influenza strains, so they are also used for influenza vaccine production.
Chinese hamster ovary (CHO) cells are a popular new type of “designed cell line”, and improvements in CHO culture technology are a major research focus.
For example, Schmitz J et al. demonstrated that MSCC benefits mammalian cell small-scale culture in terms of specific growth rate, cell diameter, and eGFP production, which is significant for the cell culture stage of vaccine production. In addition, the ActiCHO process significantly increased cell volume and monoclonal antibody titer compared to traditional processes in CHOBC clones expressing hIgG1. CHO cells have a complex glycosylation system that stabilizes SARS-CoV-2 expression and increases the content of glycosylated spike (S) protein with higher antibody detection sensitivity and specificity.
Recently, a recombinant SARS-CoV-2 protein vaccine jointly developed by the Institute of Microbiology, Chinese Academy of Sciences and Anhui Zhifei Longcom Biopharmaceutical Co., Ltd. was produced in CHO cells. Esposito et al. showed that the VRC S-protein yield exceeded 5 mg/L when captured using immobilized metal ion affinity chromatography (IMAC) columns and purified via desalting columns at 32 °C.
The human embryonic kidney cell line HEK293 is the main cell line for the transient expression of recombinant proteins. HEK293 cells were immortalized by transfection with cleaved human adenovirus type 5 (Ad5), with genes for adenoviral proteins E1A and E1B integrated into their genome. Expression of these proteins promotes HEK293 growth by regulating the cell cycle and apoptosis. Expi293F™ is also used for COVID-19 vaccine production; currently, using conventional transfection reagents and protocols, the final yield of the receptor-binding domain (RBD) is 90 mg/L.
Each virus has an optimal cell line. Human diploid cells such as MRC5 are ideal due to genetic stability and low adverse reactions in humans. Although suitable for vaccine production, human diploid cells are not amenable to large-scale culture, and cell density remains a bottleneck.
3. Development and Process Optimization of Bioreactors for Viral Production
3.1 Types of Bioreactors
Traditionally, embryonic stem cells, induced pluripotent stem cells, mesenchymal stem cells, etc., are expanded and passaged in media containing serum and feeder layers, forming a standard two-dimensional (2D) culture model. This method requires feeder layer removal but carries risks of pathogen contamination and is inconvenient. Cultured stem cells are prone to mutation, and cell yield is limited.
The use of bioreactors has partially solved these problems. Early bioreactor design was based on stirred microbial fermenters. With technological maturation, many low-shear bioreactors have been developed to overcome shear stress damage to animal cells.
The key to the large-scale industrialization and commercialization of cell culture technology is the design of appropriate bioreactors. Cell culture bioreactors are critical equipment in vaccine production, providing a suitable growth environment, determining cell quality and yield, and influencing production efficiency and product quality. Continuous multi-stage culture systems or immobilization agents can also be used for viral vaccine production.
As two-stage bioreactors, continuous bioreactors can operate under steady-state conditions (fixed cell and metabolite concentrations, pH, avoiding shutdown for cleaning and sterilization). Cell culture bioreactors can be classified into mechanically stirred, airlift, hollow-fiber, and single-use bioreactors.
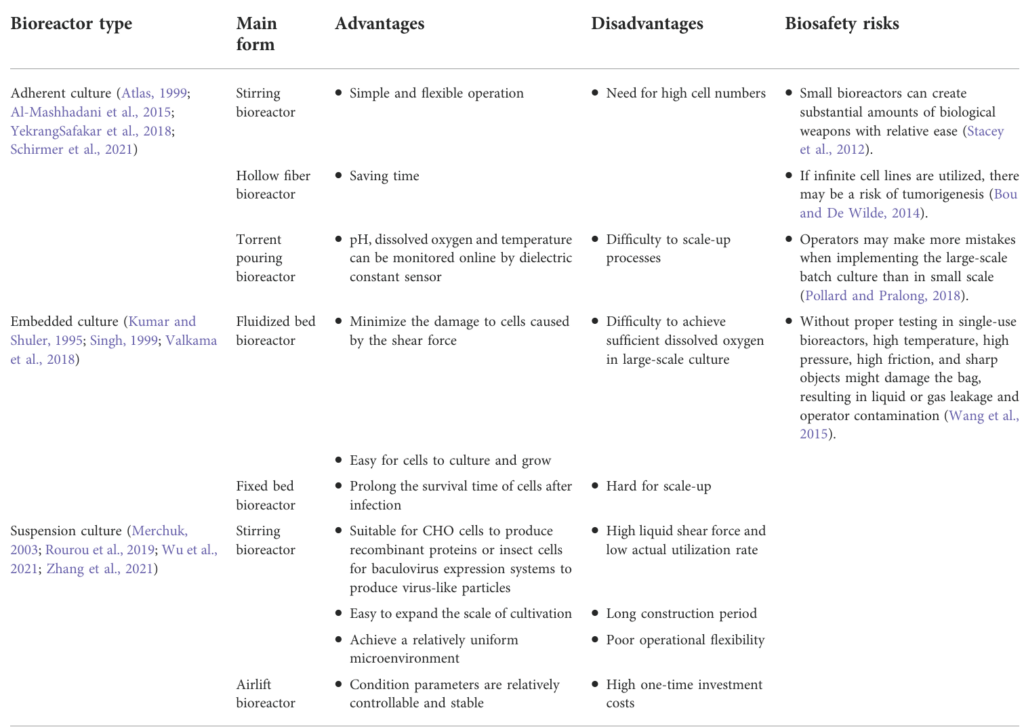
Based on culture mode, they fall into three categories: adherent, embedded, and suspension bioreactors. Their advantages, disadvantages, and biosafety risks are listed in Table 1.
In recent years, static cell culture devices of various scales have been updated. Currently, single-use fixed-bed bioreactors such as the iCELLis® 500 and 500+ systems are used for adherent cell culture to produce high-yield live viruses. Single-use fixed-bed bioreactors are simple, flexible, time-saving, and allow online monitoring of pH, dissolved oxygen (DO), temperature, and other parameters via dielectric sensors.
In the production of recombinant VSV-based live vaccines using Vero cells, the latest Univercells scale-X fixed-bed bioreactor system increased virus titers per surface area by 2–4-fold. The latest innovation in fixed-bed bioreactors is Corning’s Ascent bioreactor, which uses unique low-shear strategies to supply oxygen and nutrients to cells.
Enzymatic digestion enables cell detachment from 2D cultures, making them suitable for high-yield, scalable viral vector synthesis. For in vitro gene therapy applications, Univercells launched the competitive scale-X bio series due to the lack of mid-scale bioreactors in the iCELLis series that are more suitable for ex vivo therapies. Studies showed that lentiviral and adenoviral vectors have similar productivity in iCELLis Nano and scale-X Hydro bioreactors, but cell distribution is superior in scale-X Hydro.
In adherent culture, cells only attach to roller bottle surfaces, and scale up is limited by labor, cost, and facility space, hindering industrial translation. These limitations have driven progress in suspension culture.
Suspension culture is generally preferred for process optimization over adherent systems. For example, Cytiva’s newly developed Wave 25 bioreactor features advanced sensors and intelligent control strategies, enhancing rocking technology and reliably maintaining cell viability and improving virus yield.
Sartorius’ Ambr250 bioreactor can run up to 48 parallel bioreactors with 100–250 mL working volumes, providing a small-scale multi-module system that overcomes limitations of adherent bioreactors. Suspension culture enables large-scale production using advanced automation and will replace traditional adherent culture as the mainstream method in future large-scale manufacturing.
 3.2 Improving Virus Yield via Process Optimization
3.2 Improving Virus Yield via Process Optimization
The goal of upstream bioprocess development is to reduce overall manufacturing costs. In principle, more substrate cells lead to higher virus production. Maintaining cell lines in optimal conditions is another key factor for high virus yields.
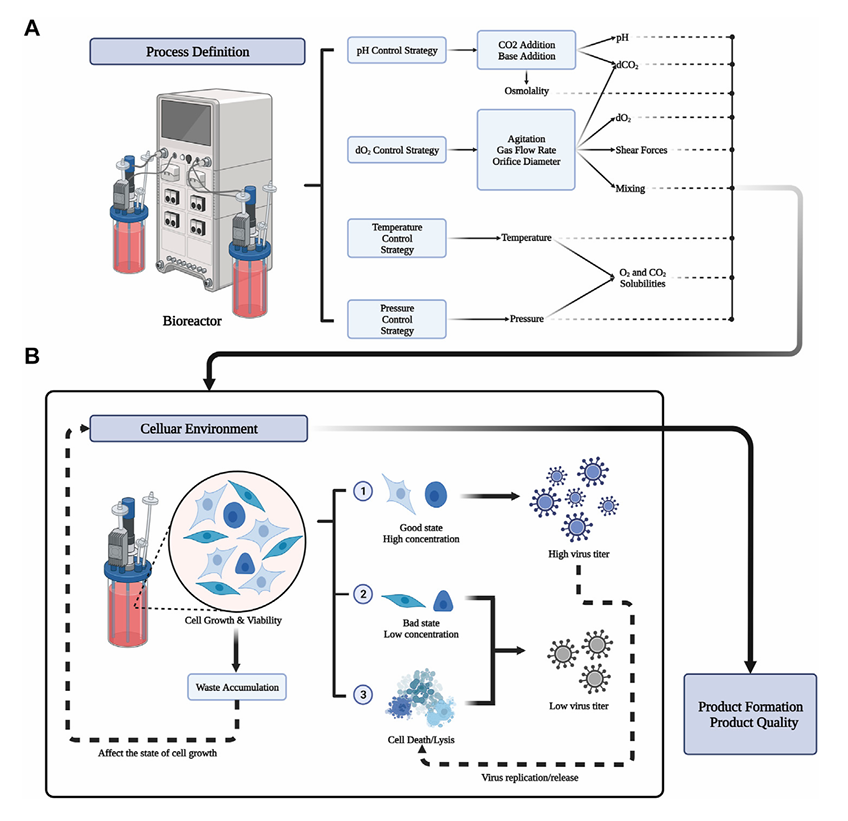
Throughout the upstream process, maintaining cells under optimal conditions for virus production is essential to maximize cellular material. This can be achieved by adjusting several parameters during manufacturing. Different process parameters in bioreactors are illustrated in Figure 2A, including physical and chemical parameters (temperature, pH, DO, osmotic pressure, shear stress, nutrient supply), multiplicity of infection (MOI), time of infection (TOI), organic nutrients, and inorganic ions.
Cell Concentration and Metabolic/Physiological State
During cell proliferation, most biological products from animal cell culture are continuously replicated and synthesized. For example, recombinant proteins are usually produced in batch or fed-batch modes and harvested once concentrations peak.
In contrast, viral vaccine production typically requires a cell growth phase followed by a viral replication phase (batch mode). Most viruses replicate through multiple infections in complex processes involving host cell, viral RNA/DNA, and viral protein synthesis, as well as progeny virus release.
Viral replication and release usually cause cell lysis, so viruses can be harvested in batches when concentrations peak. Generally, cell concentration determines final virus titer. Key nutrients and accumulated byproducts such as lactate and ammonia inhibit viral amplification. The impact of cell status on virus production is shown in Figure 2B. Reasonable parameters can be simulated in small scale cultures to provide optimal cell environments, but optimizing parameters at large scale is more costly.
Напряжение сдвига
Shear refers to the sliding of parallel layers. Shear forces generated by stirring and sparging can affect enveloped virus production in bioreactors.
For example, Grein et al. found that measles virus is highly susceptible to shear stress; stirring and sparging reduced virus titers by up to 1000-fold in some cases. Headspace aeration can provide sufficient oxygen while reducing shear effects caused by spraying.
Novel bioreactors largely overcome shear stress. Sartorius Stedim Biotech’s single-use BIOSTAT® RM TX bioreactor is equipped with single-use Flexsafe® RM TX bags for gentle agitation and dedicated ports for non-manual gravity harvesting. This unique gravity harvest concept avoids contamination risks from manual operation, reduces shear stress on fragile cells, and maximizes cell recovery.
Although shear stress is much lower in adherent culture, scale-up is difficult. The opposite is true for suspension culture. Therefore, methods to reduce shear stress in suspension culture should be further explored to improve virus yields at large scale.
Multiplicity of Infection
Viruses mainly spread to target cells through the medium, so virus degradation or inactivation before reaching target cells must be considered when calculating optimal inoculum levels. Secondary infection is critical in continuous culture.
Virus transport, spread, and re-adsorption follow complex mathematical models. In stirred-tank bioreactor cascade amplification, an optimal MOI usually exists for maximum harvestable virus titer. For most viruses, excessive viral particles per cell during infection may cause the replication of defective interfering particles, reducing maximum achievable yields.
Residence Time and Harvest Time Point
When cells or viruses are retained in a closed system, it operates in batch mode. Typically, once maximum titers are reached, infectious titers and total viral particles begin to decline. However, the most suitable harvest time is when extracellular DNA and protein contamination start to increase significantly.
Unlike classical recombinant protein production, cell growth and viral replication phases in vaccine production require different parameters, so specific process strategies must be adopted for each stage.
To achieve high productivity and low cost for viral vaccine production under steady-state conditions, optimal virus residence and harvest times must be determined. This prevents rapid virus degradation and associated yield loss, maintaining a high proportion of infectious particles, which benefits live attenuated or viral vector vaccines such as recombinant vaccinia and influenza A vaccines.
However, process control and monitoring in viral vaccine production remain unclear. Several open questions from the FDA and EMA regarding long-term genetic stability of cell and viral strains, especially whether over-production in continuous culture causes viral mutations and the negative impact of time on product efficacy and safety, still need answers.
Microcarrier Culture
Microcarrier culture is considered the most promising large-scale animal cell culture technology, combining the advantages of suspension and adherent culture. Harmless microcarrier particles are added to the medium, allowing cells to attach and grow, while continuous agitation keeps microcarriers suspended.
Cell densities of adherent cells vary due to differences in available growth area. Microcarriers are often added to bioreactors to increase cell densities. Rourou et al. showed that Vero cells grown on 3 g/L Cytodex 1 microcarriers reached 2.6×10⁶ cells/mL. However, microcarriers require high seeding densities, which may exacerbate cell density effects and hinder scale-up.
Microcarrier-based stirred-tank bioreactors are commonly used to produce and maintain many anchorage-dependent cells but may cause cytopathic effects (CPE) due to harmful hydrodynamic shear stress. As a novel solution, hollow microcarriers (HMCs) have been proposed to protect cells from shear stress while ensuring sufficient gas and nutrient supply and uniform mass transfer, facilitating large-scale amplification of shear-sensitive anchorage-dependent cells at industrial scale.
High Cell Density Virus Culture
With increasing vaccine demand, cell culture-based viral vaccine production requires technological enhancements to overcome traditional limitations. For many conventional vaccine cell lines, production cell densities range from 2×10⁶ to 4×10⁶ cells/mL, representing high cell density. Several processes and devices for high-density culture (acoustic filters, hollow-fiber systems, CellTank®, etc.) have entered the market.
Studies show that high cell densities can be achieved via improved strategies such as fed-batch and perfusion cultures. However, high-density culture reduces cell-specific virus productivity and causes cell density effects (decreased specific productivity in virus-cell expression systems at high cell densities), a common phenomenon. Perfusion technology can mitigate cell density effects by providing a continuously nutrient-rich environment and avoiding unnecessary byproduct accumulation, thus maintaining cell-specific yields.
Perfusion culture has small culture volumes, high recovery volumes, and short product residence times, enabling rapid low-temperature recovery and preservation, which benefits product activity. However, perfusion is operationally complex and has low medium utilization efficiency, possibly requiring timely medium replacement.
A study showed that after optimizing nutrient ratios, concentrations, and osmotic pressure, the resulting medium maintained 3×10⁷ cells/mL at half the perfusion rate, providing a reference for perfusion medium development. High density and viability are major features of perfusion culture, but cell density cannot increase infinitely. Excessively high density increases medium viscosity, preventing further culture.
Perfusion rates must be adjusted to maintain stable cell densities. Recent studies show that automated high-precision control of perfusion rates can be achieved by detecting cell-specific perfusion rates and substrate metabolism, achieving higher cell concentrations and viral titers up to 1×10¹⁰ viruses/mL. However, perfusion culture remains complex with low medium efficiency. Medium replacement may increase sterility risks, and systems often do not operate at optimal exchange rates.
4. Large-Scale Culture of Different Viruses
Process optimization includes cell line selection, physical and chemical parameter tuning, microcarrier technology, and high-density culture. Virus yields vary with cell lines, media, bioreactors, and physical/chemical conditions such as temperature, pH, and MOI.
Identifying optimal conditions for different viruses can greatly increase virus and vaccine yields to meet global demand. This section summarizes and reviews specific process optimizations for large-scale production of SARS-CoV-2, influenza, tropical viruses, enteroviruses, and rabies virus (Table 2).
 4.1 SARS-CoV-2
4.1 SARS-CoV-2
The COVID-19 pandemic broke out globally in 2020, posing a severe threat to public health. The WHO declared it a Public Health Emergency of International Concern. Given its severity, developing safe and effective vaccines against SARS-CoV-2 became a research priority. As of February 20, 2022, 195 vaccine candidates were in preclinical development and 144 in clinical development, mainly using four platforms: inactivated virus, protein subunit, adenoviral vector, and mRNA.With the worsening pandemic, we need more efficient bioprocesses to make enough vaccines.
Several studies talk about large‐scale SARS-CoV-2 growth in bioreactors. For example, Offersgaard’s team put 1.5×10⁷ Vero cells in 0.5 L CelCradle™ 500‐AP flasks with 5.5 g BioNOC™ II carriers. They used animal‐free medium for 7 days. Then total cells reached (2.2–2.5)×10⁹ per flask. They used MOI 0.006 at 33 °C. The peak infectious titers reached 7.3 log₁₀ TCID₅₀/mL at 72 hpi. Six harvests were ≥6.5 log₁₀ TCID₅₀/mL. The total was 10.5 log₁₀ TCID₅₀ in about 5 L.
BBIBP‐CorV was the world’s first inactivated SARS-CoV-2 vaccine. Researchers compared it with CelCradle™ 500‐AP. They used a new carrier‐based way in basket bioreactors. This method supports efficient production. They used MOI 0.01–0.3. Titers reached 7.0 log₁₀ CCID₅₀/mL at 48–72 hpi.
In solid fluidized‐bed systems, cells stay relatively still. This makes high‐density culture easier. Wang et al. tested three candidate strains. HB02 gave the highest yields in Vero cells. It had no amino acid change within 10 passages. So virus strain type strongly affects the final yield.
ChAdOx1 nCoV-19 (AZD1222, Vaxzevria) is an effective adenoviral vector COVID-19 vaccine. The virus is made in HEK293 cells. Joe et al. grew HEK293 cells in BalanCD HEK293 medium and Pall Allegro bioreactors. The cell density reached (2.5–3.0)×10⁶ cells/mL. They used MOI 5–10. Adenoviral vectors reached 5×10¹¹ viral particles (VP)/mL at 42–48 hpi. This team completed one of the largest viral vector production projects so far. It supplied many global COVID-19 vaccines at a low cost. Its productivity was about two times higher than old batch or fed‐batch methods. Perfusion culture works well for mammalian cells and adenoviral vectors.
Scientists are still studying rVSV‐based SARS-CoV-2 vaccines. Kiesslich et al. compared production in MDXK and IHM03 media. They grew Vero cells in MDXK at 31 °C, pH 7.2, DO 50%. Cell density reached 1.02×10⁶ cells/mL. They used MOI 0.01. Titers reached 3.59×10⁹ TCID₅₀/mL and 2.13×10¹⁰ viral genomes (VG)/mL at 48 hpi.
Recombinant protein COVID-19 vaccines, such as ZF2001 and NVX‐CoV2373, are now available. These vaccines focus on antigens, not whole viruses. They often use engineered CHO cells, such as Epi CHO, CHO Freestyle Max, and CHO‐S. Johari et al. grew CHO‐S cells in CD‐CHO medium with 8 mM L‐glutamine at 37 °C. They used suspension culture. When density reached 1.5×10⁶ cells/mL, they treated cells with PEI. They transfected cells with a vector that has the spike gene and a 100 RPU promoter. They cultured these cells at 32 °C in fed‐batch mode. They got 53 mg/L purified spike protein.
In a similar way, Pino et al. grew codon‐optimized CHO Express™ cells in EX‐CELL® Advanced™ CHO medium at 37 °C. They used suspension culture. They transfected cells with pXLG6 that has SARS‐CoV‐2 spike DNA. After 10 days, they moved the culture to 50 mL Tube Spin® bioreactors. They used fed‐batch production at 31 °C. Then they scaled up to 10 and 40 L stirred tanks. Yields increased greatly. The CHO expression system gives high yields and can be scaled up.
Vero cells are widely used for SARS‐CoV‐2 because they are very sensitive. CHO cells that make viral proteins are also a main research focus. More new bioreactors are used for SARS‐CoV‐2 than for other viruses. But researchers still need to improve environment monitoring and medium optimization during the whole culture process.

4.2 Influenza Virus
Influenza A virus is a big global health threat, just like SARS‐CoV‐2. Vaccines are the most effective and safe way to prevent it. Scientists have built cell‐culture‐based processes. They use MDCK, Vero, AGE1.CR, or PER.C6 cells.
For example, Lai et al. used a new mammalian cell line called PBG.PK2.1. They also used a hollow‐fiber ATF system (ATF2) for high‐density culture. Cell density reached 50×10⁶ cells/mL. The maximum HA titers were 3.93 log₁₀ HAU/100 mL. PBG.PK2.1 cells can reach high concentrations. They work with chemically defined media. They are also easy to scale up. So they are good candidates for vaccine production.
They also did two perfusion cultures in bioreactors. The cell density at infection was about 4.6×10⁶ cells/mL. The maximum titer was (3.93±0.05) log₁₀ HAU/100 mL. This shows that perfusion improves production and allows continuous virus collection.
Many studies now prefer perfusion technology. Bissinger et al. grew MDCK cells in suspension with semi‐perfusion mode. They got 4.5 log₁₀ HAU/100 mL and 10¹⁰ viral particles/mL at MOI 10⁻¹. In a later study, they scaled semi‐perfusion to bioreactors with ATF perfusion. They adjusted pH and temperature. The cumulative HA titers (HAaac) reached 4.37 log₁₀ HAU/100 μL, which is 4.7×10¹¹ viruses/mL.
ATF systems affect virus production. Coronel et al. used SB10‐X orbitally shaken bioreactors (OSB) with TFF and ATF. Under the best perfusion conditions, CSVY reached 3500 viral particles per cell. Volumetric productivity (Pv) was 2.2×10¹² particles/L/d. The maximum HA was 3.73 log₁₀ HAU/100 mL. TCID₅₀ was 8.8×10⁹ infectious particles/mL.
Inclined settlers (IS) gave more viruses than ATF. The total was (5.4–6.5)×10¹³ particles. In IS 1, trypsin activity was at its highest level. The value was 1.5×10⁶ U/cell or 38 U/mL. All cells were infected at 24 hpi. When the infection density was 25×10⁶ cells/mL, cell‐specific virus productivity reached 3474 particles per cell. This greatly improved IAV yields and helped influenza vaccine production.
Researchers can improve perfusion technology for influenza A in two ways. They can use new perfusion bioreactors, or they can change physical and chemical parameters. Perfusion is easy to scale up. It is very suitable for making more influenza vaccines every year.

4.3 Tropical Viruses
Dengue is a virus infection spread by mosquitoes. It has four serotypes: DENV‐1, ‐2, ‐3, and ‐4. Only Dengvaxia® is licensed right now. Serum‐free media have been used for many years. Liu CC et al. found that all four DENV serotypes grew better in Vero or MRC‐5 cells in SFM. Titers were 0.3–2.6 times higher than in serum‐containing media. Vero cells gave peak titers that were 1–17 times higher than MRC‐5 cells. But Lee HC’s group confirmed that DENV‐4 from MRC‐5 cells is more stable. MRC‐5 cells grown on microcarriers are genetically more stable than Vero cells. But it is still hard to scale them up.
Zika virus (ZIKV) belongs to the Flaviviridae family. Mosquitoes mainly spread it. The choice of cell line and bioreactor strongly affects production. Nikolay et al. made ZIKV from BHK‐21 suspension cells in DMEM. They used 3 L single‐use Mobius® bioreactors at pH 7.1.
They wanted to improve titers. So they used ATF perfusion. They got 3.9×10⁷ PFU/mL at 4 dpi with MOI 0.001. Cell‐specific virus yields were still lower than in adherent culture. But they built a large enough ZIKV production process.
Perfusion technology kept improving. They used hollow‐fiber‐based perfusion in bioreactors with ATF/TFF. This helped cell growth and increased titers. They made the virus in EB66® cells. These cells grew in GMEM at 34 or 37 °C. They got 1.0×10¹⁰ PFU/mL at 2 dpi with MOI 0.001. The best seeding density was 1.6×10⁸ cells/mL. They used capacitance probes to automatically control perfusion rates based on online cell density measurements. They successfully automated the whole process.
Total yields became 1000 times higher than older studies. This came from high‐density culture, EB66‐adapted seed virus infection, and hollow‐fiber perfusion.
Chikungunya virus (CHIKV) is an alphavirus spread by mosquitoes. It has infected millions of people around the world. Scientists have successfully produced it in insect cell lines. Pijlman et al. optimized DASGIP® bioreactors. They found the best conditions for BACe56‐CHIKV recombinant virus. MOI was 0.01 TCID₅₀. Seeding density was 2×10⁶ cells/mL. Harvest time was about 52 hpi. They got 2.1 mg/L CHIKV VLPs.
Another study made CHIKV in Vero cells. Ramya et al. grew cells in bioreactors to 4×10⁵ cells/mL. Then they added the virus. They finally got 1.4×10⁹ PFU/mL. Most current research focuses on recombinant CHIKV vaccines. Few studies look at bioreactor optimization. Insect cells give more choices for upstream production.
The best vaccine candidate for Ebola virus is rVSV‐ZEBOV. Gélinas et al. used a 3.5 L stirred‐tank bioreactor. They grew HEK293SF cells in HyClone HyCell TransFx‐H medium at 34 °C. They infected cells at MOI 0.001. The cell count was 1.16×10⁶ cells/mL at infection. It reached 1.8×10⁶ at harvest. Titers reached 1.19×10⁸ TCID₅₀/mL and 50 TCID₅₀/VG at 36 hpi.
Kiesslich et al. used a new scale‐X hydro fixed‐bed bioreactor to make rVSV‐ZEBOV. They used VP‐SFM medium at 34 °C and DO 50%. The maximum cell density reached 271605 cells/cm². They used MOI 0.01. The peak infectious titers reached 1.95×10⁷ TCID₅₀/mL at 24 hpi. The VG/TCID₅₀ ratio was only 101.
Cell‐specific infectious particle productivity became 1.9 times higher than in microcarrier systems. But total particle productivity per cell dropped 5.9 times. They wanted to improve titers. So they made rVSV‐ZEBOV in suspension Vero cells in IHM03 and MDXK media. In IHM03 with MOI 0.01, titers reached 1.05×10⁷ TCID₅₀/mL. At 4×10⁸ cells/mL, titers reached 1.32×10⁸ TCID₅₀/mL.
Suspension‐adapted Vero cells worked better than MDCK cells. Cell density reached 1.02×10⁶ cells/mL. The maximum titer was 3.87×10⁷ TCID₅₀/mL. It reached 37.9 TCID₅₀ per cell. The VG/TCID₅₀ ratio was 282. New and better bioreactors helped rVSV‐ZEBOV yields and infectious titers increase a lot.

4.4 Enteroviruses
Enterovirus 71 (EV71) causes hand, foot, and mouth disease (HFMD). China launched the EV71 vaccine in 2016. It prevents HFMD caused by EV71. Current EV71 vaccine candidates are made in Vero cells. Researchers use roller bottles or cell factories because these methods are easy to use.
These methods need a lot of labor. So we need more efficient and cheaper processes. Microcarrier bioreactors support large‐scale production and higher titers. Chen et al. used a new mini‐bioreactor with polymer fiber carriers. They used MOI 0.1 at 32 °C. The peak virus titer reached 1.0×10⁸/mL at 3 dpi. The total supernatant volume was 25 L. The total virus amount was 1.93×10¹². They built a new EV71 production model. It supports fast and low‐cost HFMD vaccine production.
Cytodex 1 microcarriers are widely used for EV71 production. Liu et al. used microcarrier combined bioreactors to increase EV71 vaccine yields. Multi‐harvest semi‐batch (MHSBC) or perfusion culture increased EV71 yields 7–14 times compared to single‐batch culture. Perfusion worked better than MHSBC. Changing medium in a smart way slowed CPE and increased EV71 virus yields.
Wu et al. used a 200 L serum‐free microcarrier bioreactor system. They set conditions at 37 °C, pH 6.8–7.2, MOI 10⁻⁴. Virus titers reached 10⁷ TCID₅₀/mL at 10 dpi. They built a large‐scale platform for inactivated EV71 vaccine production.
These studies give useful information for large‐scale microcarrier cell culture processes. These processes make inactivated EV71 vaccines in Vero cells. The choice of medium is very important for the final yield. Scientists tested VP‐SFM during upstream development. It is the best medium for Vero cell growth and EV71 production in pilot vaccine production.
Coxsackievirus A16 (CVA16) is another main cause of HFMD. There is no safe and effective licensed CVA16 vaccine yet. The situation is more complex. EV71 virus particles do not create cross‐reaction neutralizing antibodies against CVA16. So we need to develop CVA16 vaccine candidates. These vaccines should be based on whole virus production in Vero cells.
Large‐scale virus culture technology has many advantages. Chen et al. grew Vero cells on polymer fiber carriers. They used serum‐free medium with 0.5% (w/w) whey protein hydrolysate. They used single‐use Bioflo310 bioreactors and amprotein current perfusion bioreactors. They monitored virus infection and Vero cell growth.
After these improvements, the final virus titer reached 7.8×10⁷ TCID₅₀/mL. This study shows that fiber carriers have low shear rate and friction. They reduce the cytopathic effect of the virus on cells. So they are better than other microcarriers. These studies will also help large‐scale production of inactivated CVA16 vaccines. These vaccines use cells grown on nonwoven polymer fiber sheets.Liu et al. increased EV71 vaccine yields via microcarrier-integrated bioreactors. Multi-harvest semi-batch (MHSBC) or perfusion culture increased EV71 yields 7–14-fold compared to single-batch. Perfusion was superior to MHSBC, and medium replacement slowed CPE and improved yields.
Wu et al. used a 200 L serum-free microcarrier system at 37 °C, pH 6.8–7.2, MOI 10⁻⁴, reaching 10⁷ TCID₅₀/mL at 10 dpi and establishing a large-scale inactivated EV71 platform.
These studies provide valuable information for large-scale microcarrier Vero cell processes for inactivated EV71 vaccines. Medium choice is a rate-limiting step; VP-SFM developed upstream is optimal for Vero growth and EV71 production in pilot-scale candidates.
Coxsackievirus A16 (CVA16) is another major HFMD pathogen, with no safe and effective licensed vaccine. EV71 particles do not induce cross-neutralizing antibodies against CVA16, necessitating CVA16 vaccines based on whole-virus production in Vero cells.
Leveraging large-scale viral culture, Chen et al. cultured Vero cells on polymer fiber carriers in serum-free medium with 0.5% whey protein hydrolysate, monitoring infection and culture in single-use Bioflo310 and ammonia-protein current perfusion bioreactors.
Final titers reached 7.8×10⁷ TCID₅₀/mL. Low shear and friction of fiber carriers reduce viral CPE, making them superior to other microcarriers. These studies will facilitate large-scale inactivated CVA16 vaccine production using cells grown on nonwoven polymer fiber sheets.
4.5 Rabies Virus
Rabies is a viral zoonosis. Dogs are the main reservoir, causing 99% of human rabies deaths. Cell-culture-derived rabies vaccines are among the safest and most effective for humans. Vero cells are widely used. High-density cells in bioreactors enable continuous virus harvest via perfusion. For example, Rourou et al. studied perfusion in 2 L bioreactors, reaching 5×10⁶ cells/mL and peak titers of 1.38×10⁸ focus-forming units (FFU)/mL, with mixed inactivated virus titers of 2.58 IU/mL.
Recently, Rourou further cultured rabies virus in suspension Vero cells. A serum-free system for suspension-adapted Vero (VeroS) was established in shake flasks at 37 °C, 5% CO₂. The LP-2061 strain infected VeroS at MOI 0.1 and (8±0.5)×10⁵ cells/mL, with all media yielding >10⁷ FFU/mL.
Cell-specific virus productivity slightly increased compared to adherent culture, confirming VeroS suitability for high-titer rabies virus production and paving the way for VeroS-based bioreactor manufacturing. However, titers from VeroS remain lower than adherent cultures.
Thus, Vero suspension technology is immature and requires further density improvements. Downstream process optimization, including physical/chemical parameters, and research on human diploid cells (HDCs) for large-scale rabies vaccine production should also be strengthened.
5. Computational Biology Simulation for Large-Scale Upstream Process Development
From laboratory simulation to industrial production, upstream manufacturing inevitably involves differences, most intuitively bioreactor scale (Figure 1A). Maintaining long-term cell stability in bioreactors ≥10,000 L is the biggest challenge in current upstream culture.
Direct large-scale optimization is unrealistic. Applying computational biology to upstream production largely solves this dilemma. Computer simulation of the entire culture process, testing and optimizing parameters to establish optimal models, can theoretically enable low-cost, high-efficiency industrial upstream manufacturing.
Ueki et al. designed an inverted mixing bioreactor with interactive mixing characteristics. They used computational fluid dynamics (CFD) software (FLUENT) to calculate shear stress and vectors for stirred and rotating bioreactors, using unsteady and steady-state analyses, respectively, and established a κ-ε model for comparison.
Results showed that maximum and average shear stresses were significantly lower in inverted mixing bioreactors than in traditional rotating ones, reducing cell damage, improving physiological activity, and increasing maximum cell density.
East China University of Science and Technology, Tongji University, and the University of Surrey proposed high-dimensional model representation (HDMR) to analyze and simulate the global sensitivity of perfusion bioreactors. Using Contois parameters, lactate Michaelis constants, glucose and oxygen maximum consumption rates and concentrations, and cell death kinetics as inputs, and glucose, oxygen, lactate, cell density, and growth as outputs,
they established comprehensive mathematical models and simulated them using COMSOL Multiphysics v5.5. HDMR provides suitable physical/chemical parameters for cell growth. Another team focused on cell cycle-environment interactions, establishing an agent-based modeling (ABM) framework to simulate single mammalian cell growth and cycle regulation in response to culture conditions. This model predicts growth via input parameters, with reliability verified experimentally.
During the COVID-19 pandemic, mathematical models were developed to simulate adenoviral vaccine production. Ferreira et al. used SuperPro Designer v12 to design, model, and economically evaluate upstream production of SARS-CoV-2 vector vaccines.
Results showed perfusion produced 1×10¹² VP/mL, yielding 4 million doses annually. Applying computational biology theoretically demonstrated large-scale, low-cost adenoviral vaccine production. However, early and late development costs were not integrated, a major limitation. Still, it provided valuable information and accelerated adenoviral vaccine development.
In summary, computer simulation of upstream culture and full vaccine production is expected to become a mainstream future direction. Although computational models minimize R&D resource consumption, current technologies have many defects, causing deviations between theoretical and actual values.
The biggest obstacles to engineering-scale bioreactor computational models are the lack of quantitative data and physical models. Ultimately, human understanding of cells remains limited. Despite achievements in simulating viral production and cell culture, many models only reconstruct known experimental phenomena, often ignoring complex cellular structures to directly simulate states, leading to significant theoretical deviations in real environments. Thus, computational biology and bioreactors require further coordinated development to address challenges from lab-to-industry scale differences.
6. Conclusion
Vaccine supply is critical for timely immunization, especially in low-and middle-income countries. Laboratory-scale research provides extensive data to support process and bioreactor selection for highly process-intensive viral vaccine production.
To further optimize large-scale viral vaccine manufacturing, these data must be integrated with novel technologies such as medium optimization, new culture protocols, high-efficiency cell lines, and automation. Recently developed omics technologies (metabolomics, genomics, proteomics) may help design high-density growth and viral generation strategies. Feed or perfusion rates can be directly controlled via online monitoring of substrate and metabolite concentrations.
In addition, advances in computational biology have enabled software-based bioreactor system modeling, simulation, and economic evaluation of vaccine production, allowing faster vaccine development against emerging outbreaks. Thus, the rapid development and maturation of these technologies are critical for efficient, low-cost vaccine production and represent important future research directions. Связаться с нами


